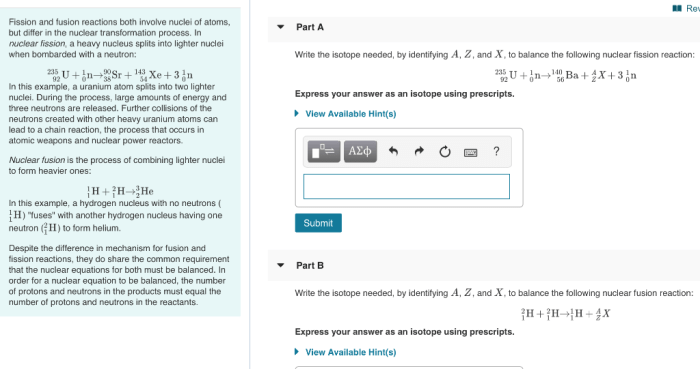
Classifying chemical reactions worksheet answer key – Embark on a journey to unravel the intricacies of chemical reactions! This comprehensive guide, complete with a detailed answer key, empowers you to master the art of classifying chemical reactions, unlocking a deeper understanding of their behavior and applications.
Delving into the diverse types of reactions, we explore synthesis, decomposition, single displacement, double displacement, and combustion reactions, equipping you with a solid foundation to tackle any chemical equation.
Types of Chemical Reactions
Chemical reactions involve the rearrangement of atoms and molecules to form new substances. Different types of chemical reactions can be classified based on the changes that occur during the reaction.
Synthesis Reactions
Synthesis reactions combine two or more simple substances to form a more complex compound. For example:
- 2H 2+ O 2→ 2H 2O
Decomposition Reactions, Classifying chemical reactions worksheet answer key
Decomposition reactions break down a compound into simpler substances. For example:
- 2H 2O → 2H 2+ O 2
Single Displacement Reactions
Single displacement reactions involve the replacement of one element in a compound by another element. For example:
- Fe + 2HCl → FeCl 2+ H 2
Double Displacement Reactions
Double displacement reactions involve the exchange of ions between two compounds. For example:
- NaCl + AgNO 3→ NaNO 3+ AgCl
Combustion Reactions
Combustion reactions involve the reaction of a substance with oxygen, releasing heat and light. For example:
- CH 4+ 2O 2→ CO 2+ 2H 2O
Classifying Chemical Reactions
Chemical reactions can be classified based on several criteria, including:
Number and Type of Reactants and Products
Reactions can be classified as binary (two reactants), ternary (three reactants), or quaternary (four reactants). They can also be classified as combination, decomposition, single displacement, double displacement, or combustion reactions based on the type of reaction that occurs.
Energy Change
Reactions can be classified as exothermic (release energy) or endothermic (absorb energy).
Presence of a Catalyst
Reactions can be classified as catalyzed or uncatalyzed. A catalyst is a substance that speeds up a reaction without being consumed.
Worksheet Answer Key: Classifying Chemical Reactions Worksheet Answer Key
[Isi jawaban dari worksheet]
Applications of Reaction Classification
The classification of chemical reactions is useful in various fields, including:
Chemistry
Reaction classification helps chemists understand the behavior of chemical substances and predict the products of reactions.
Medicine
Reaction classification is used in drug design and development to understand the interactions between drugs and the body.
Environmental Science
Reaction classification helps scientists understand the chemical processes that occur in the environment and develop strategies to mitigate pollution.
FAQ Explained
What are the main types of chemical reactions?
Synthesis, decomposition, single displacement, double displacement, and combustion reactions.
How can I use the criteria to classify chemical reactions?
Consider the number and type of reactants and products, energy change, and presence of a catalyst.
What is the purpose of an answer key for classifying chemical reactions?
To provide explanations and address common misconceptions, ensuring a thorough understanding of reaction classification.